HPLC/UHPLC Technical Tip
Level: Basic
Understanding the van Deemter Equation in Chromatography - Part 1
The van Deemter equation, developed in 1956, provides a theoretical framework for understanding band broadening in chromatographic columns. Band broadening refers to the spreading of a molecular species as it moves through a chromatography column, resulting in a broader peak on the chromatogram instead of an ideal narrow peak.
Thus, this equation serves as a guide to understanding and optimizing the efficiency of separation, leading to proper and precise analytical results. Application of the van Deemter equation can ensure sharp and clear-cut separation of analytes. This equation helps in the following:
• Determining the optimal flow rate of the mobile phase
• Minimizing band broadening
Components of the van Deemter Equation
The van Deemter equation is fundamental in chromatography, elucidating how various factors contribute to peak broadening and, consequently, affect the efficiency of a chromatographic column. The equation is expressed as:
H = A + B/u + C•u
Where:
• H represents the height equivalent to a theoretical plate (HETP), a measure of column efficiency.
• A is the eddy diffusion term.
• B/u is the longitudinal diffusion term.
• C•u is the mass transfer term.
• u denotes the linear velocity of the mobile phase.
Eddy Diffusion Term (A): This term accounts for the multiple pathways that analyte molecules can traverse through a packed column. Due to the non-uniform size and arrangement of packing particles, molecules may follow paths of varying lengths, leading to peak broadening. Minimizing this effect involves using uniformly packed, small, spherical particles, which reduce the eddy diffusion contribution.
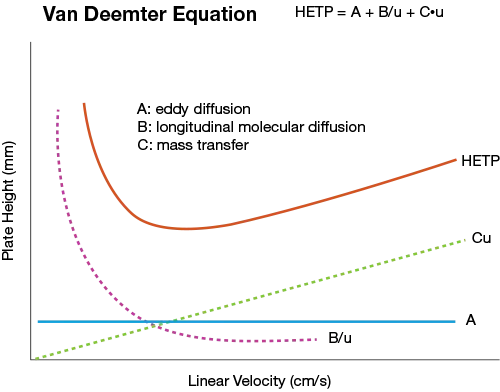
Longitudinal Diffusion Term (B/u): This term describes the natural tendency of analyte molecules to diffuse longitudinally along the column axis due to concentration gradients. Such diffusion can cause peak broadening, especially at low mobile phase velocities, as molecules have more time to disperse. To mitigate longitudinal diffusion, higher flow rates are employed, reducing the time analytes spend in the column and limiting diffusion.
Mass Transfer Term (C·u): This term represents the resistance to mass transfer between the stationary and mobile phases. Analyte molecules may interact differently with these phases, leading to variations in migration speeds and peak broadening. Factors influencing this term include the thickness of the stationary phase and the diffusion coefficients of the analytes. Optimizing these parameters can enhance mass transfer and improve separation efficiency.
The ultimate goal of this equation is to find the flow rate where the factors affecting the efficiency of chromatography are minimized. Further, the van Deemter equation acts as a balancing tool for determining better resolution between closely eluting compounds, obtaining sharper peaks, and reducing the analysis time without sacrificing accuracy.
Variations to the van Deemter Equation
Variations in the van Deemter equation arise due to changes in column design or operational conditions. These variations can affect separation efficiency.
• The B term in the van Deemter equation is influenced by diffusion coefficients in the mobile, stagnant mobile, and stationary phases. The variations in these phases may arise due to the medium's properties. For instance, diffusion rates in the octadecyl-silica (ODS) stationary phase are about half those in methanol-water mixtures, significantly affecting the B term. Moreover, the higher viscosity of the mobile phase suppresses longitudinal diffusion.
• High-pressure conditions in chromatographic columns create longitudinal temperature and pressure gradients, altering diffusion coefficients along the column. This leads to a modified van Deemter equation, replacing the classical linear C-term with more complex forms to account for these axial gradients.
• The original van Deemter equation assumptions can misrepresent mass transfer mechanisms, especially in hydrophilic interaction liquid chromatography (HILIC) and reverse phase liquid chromatography (RPLC). Adjustments are needed to incorporate accurate diffusion coefficients and mass transfer resistances for both mobile and stationary phases.
• In gas chromatography, carrier gas pressure impacts the van Deemter equation parameters, particularly the B term. Models like the Giddings model account for pressure drops, highlighting the need to carefully consider pressure effects to avoid overestimating diffusional smearing and ensure accurate parameter values.
We will cover the factors affecting the van Deemter equation and how to optimize chromatographic performance with the equation in a future technical tip. Stay tuned!
HPLC Knowledge Center
Deepen your understanding of high-performance liquid chromatography (HPLC). Our Knowledge Center offers a wide range of learning materials tailored to your needs. Dive into webinars, educational videos, insightful blog articles, and a comprehensive application library.
The Chromatographer's Guide to Improving UHPLC Column Selectivity
How to choose the most effective UHPLC column selectivity for your application.
Phenomenex, the P icon, and the Phenomenex product and service marks mentioned herein are trademarks or registered trademarks of Phenomenex, Inc. in the United States and/or other countries. All other trademarks are the property of their respective owners. One or more authors are affiliated with Phenomenex, Inc. FOR RESEARCH USE ONLY. Not for use in clinical diagnostic procedures. © 2026 Phenomenex, Inc. All rights reserved.